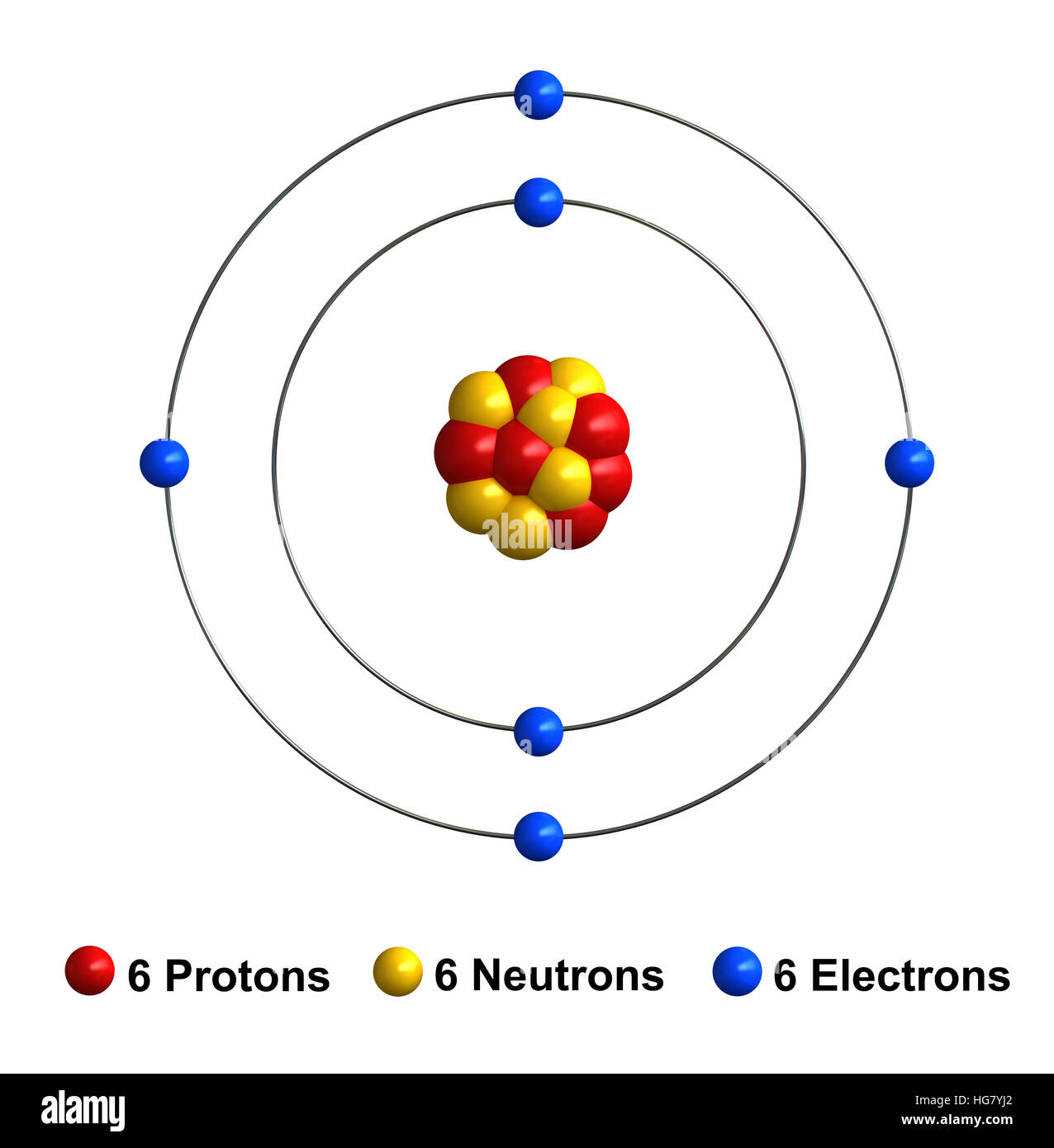
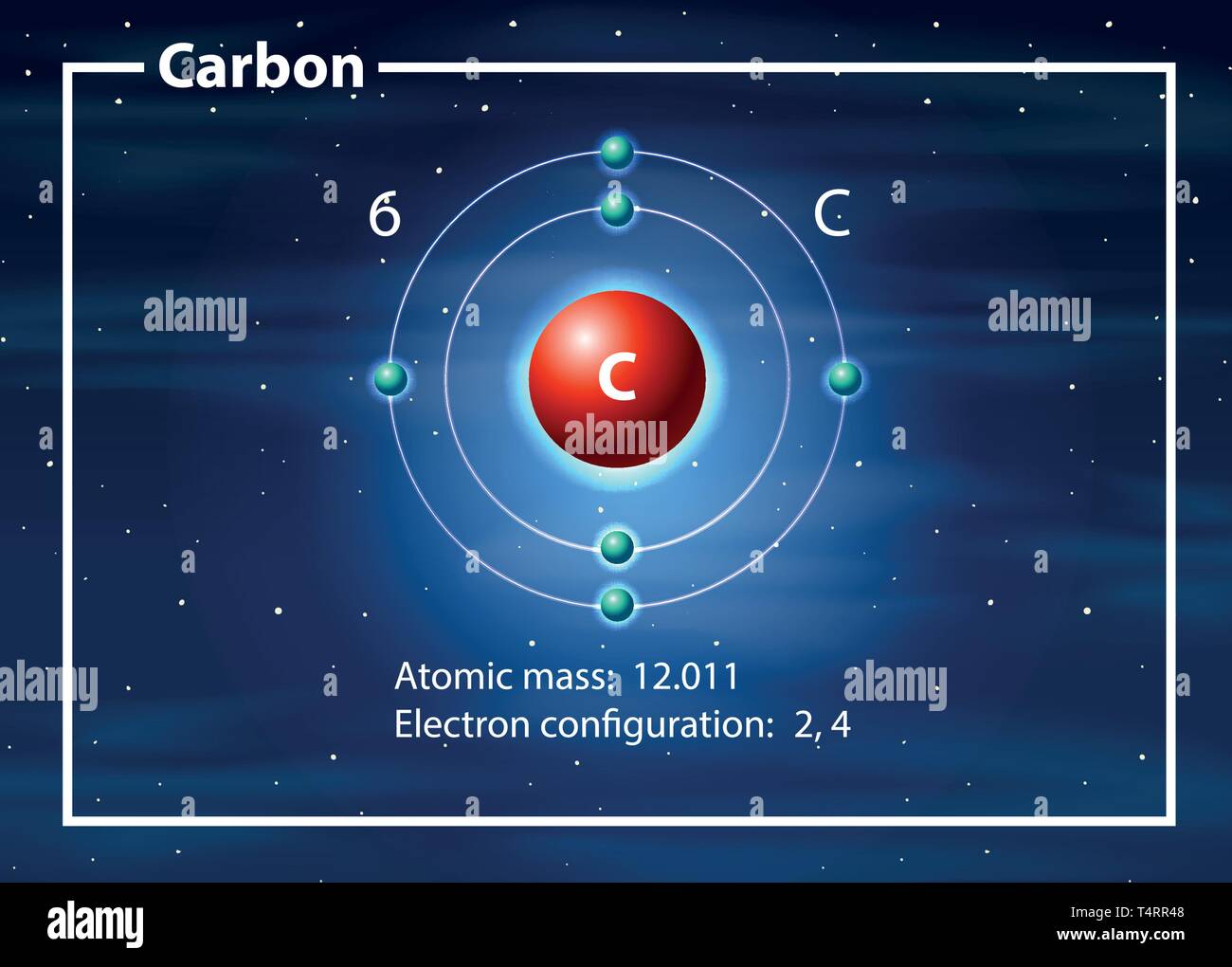
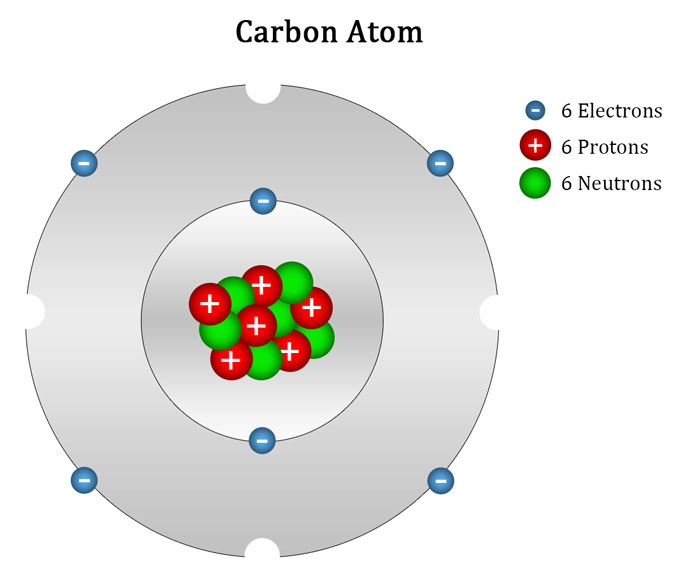
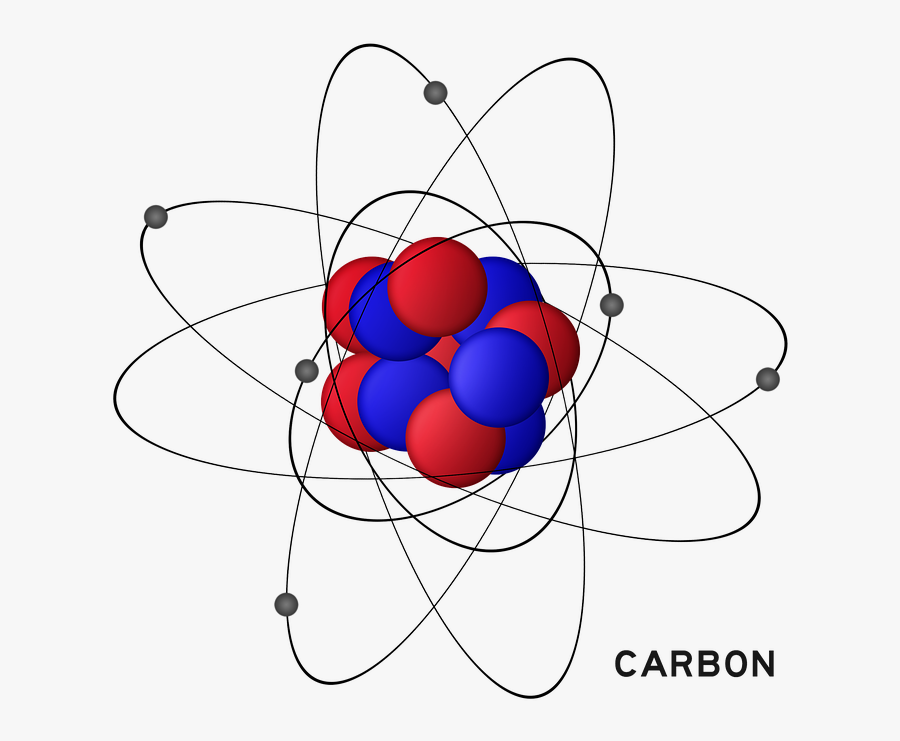
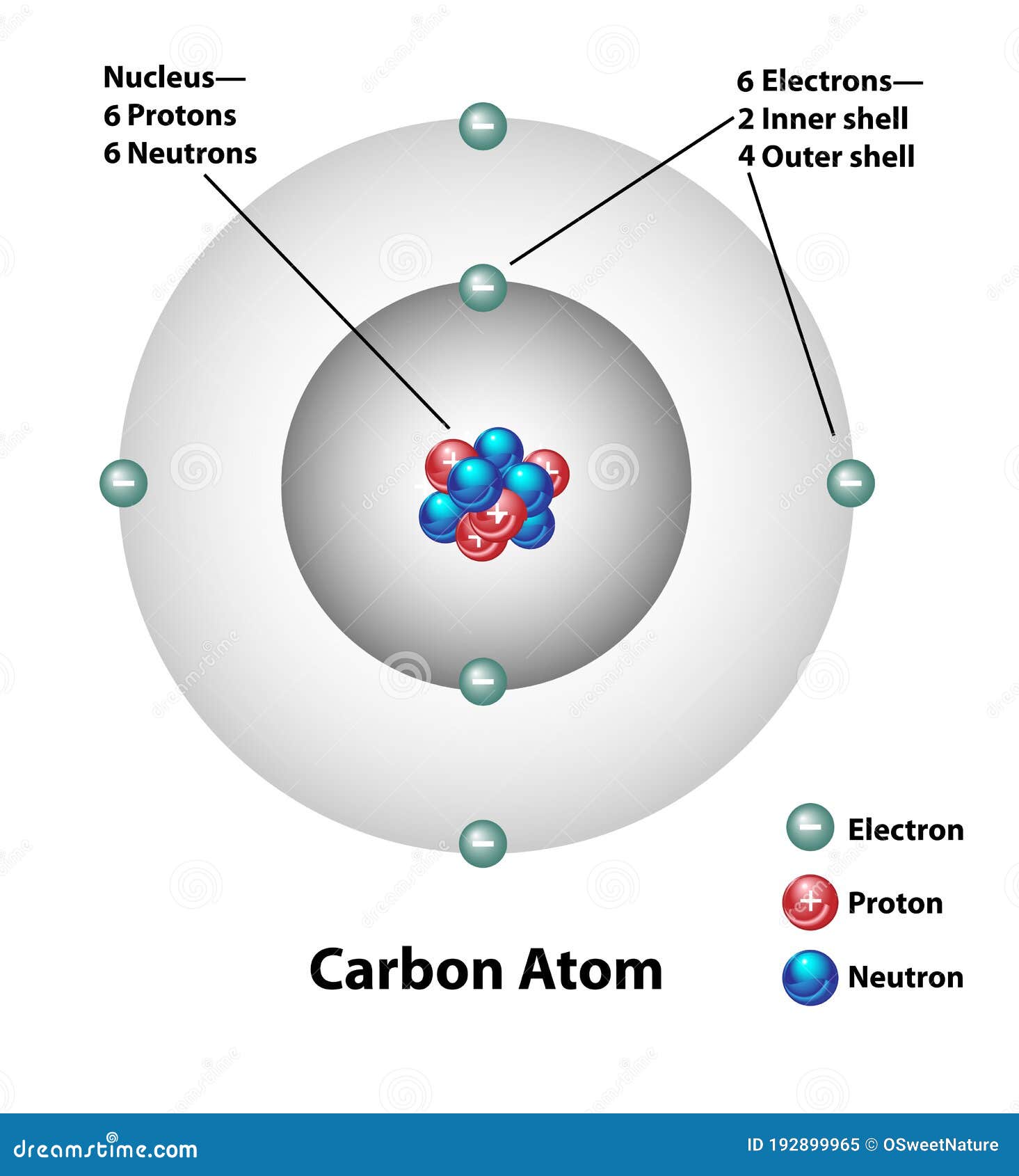
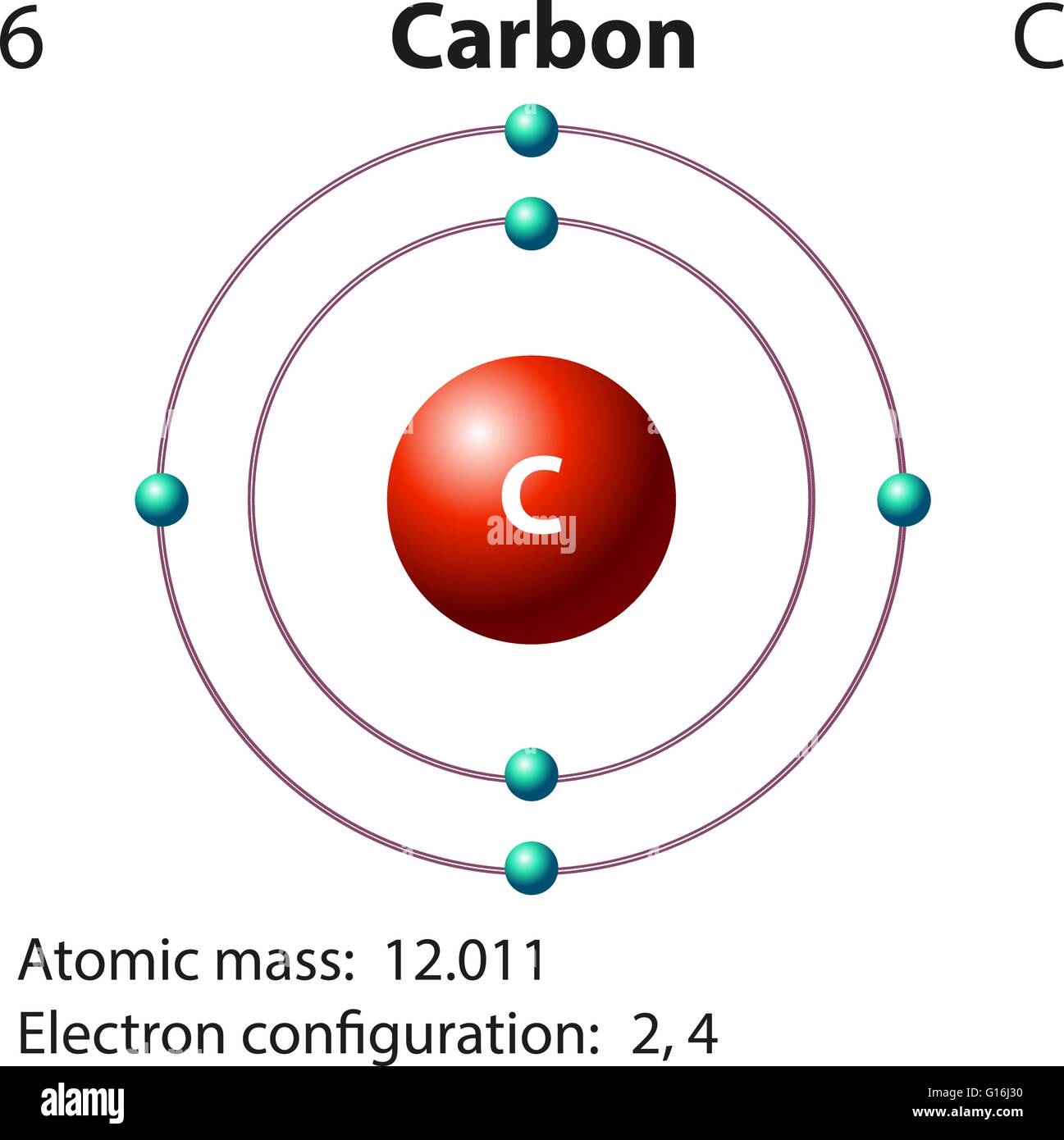
Carbon Atom Drawing
Carbon Atom Drawing - Web this image of a carbon atom is taken from a postsecondary chemistry textbook. Oxygen is more electronegative than carbon. It is nonmetallic and tetravalent —meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. Web when drawing the structures, writing out each carbon atom, with all attached hydrogen atoms, can become long and difficult. And let's look at our periodic table to figure out the relative values for those. Carbon typically shares electrons to achieve a complete valence shell, forming bonds with multiple other atoms. All structures must begin with an atom. It belongs to group 14 of the periodic table. Carbon is the first element that has precisely this shape, it is made up of 12 protons in a icosahedron shape. Web we will use this information to draw the bohr model of the carbon atom. Converts the structural formula into a 3d model. The quantum mechanical model of the atom. In the first step, we will draw the nucleus of the carbon atom. Web the small grey dot that follows your cursor is an atom (carbon). It belongs to group 14 of the periodic table. Web this image of a carbon atom is taken from a postsecondary chemistry textbook. We’ll use a bohr diagram to visually represent where the electrons are around the nucleus of the c. Web drawing every bond and every atom is tedious, however, so chemists have devised several shorthand ways for writing structures. It is nonmetallic and tetravalent —meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. Web explore the interactive simulation to build and understand atoms, isotopes, and their periodic table representations at phet. Web a carbon atom is present wherever a line intersects another line. And let's look at our periodic table to figure out the relative values for those. Web lewis structure of carbon dioxide: Hover and click on a placed atom to place a bond. Create a chain of carbon atoms. Carbon is the first element that has precisely this shape, it is made up of 12 protons in a icosahedron shape. Hydrogens that are attached to elements other than carbon are shown. Web so if we ignore hydrogen, the central atom is either going to be carbon or oxygen. Web the most common carbon atoms have six protons and six. The quantum mechanical model of the atom. This figure explains the bonding in a co2 molecule. Web the small grey dot that follows your cursor is an atom (carbon). It is nonmetallic and tetravalent —meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. As long as it’s carbon. Web drawing every bond and every atom is tedious, however, so chemists have devised several shorthand ways for writing structures. An isotope of uranium has an atomic number of 92 and a mass number of 235. What are the atomic number and the mass number of these carbon atoms? Web we will use this information to draw the bohr model. Converts the structural formula into a 3d model. A system was developed to shorthand the chemical compounds. Web in this video we'll look at the atomic structure and bohr model for the carbon atom (c). Carbon is the first element that has precisely this shape, it is made up of 12 protons in a icosahedron shape. The full page where. It belongs to group 14 of the periodic table. Quantum numbers for the first four shells. Pick one of the bond types (single, double, triple, up, down) and add or modify bonds. The atomic number symbolizes the number of protons in an atom. Converts the structural formula into a 3d model. Web we will use this information to draw the bohr model of the carbon atom. All structures must begin with an atom. This figure explains the bonding in a co2 molecule. The full page where it appears can be seen below. Carbon is the first element that has precisely this shape, it is made up of 12 protons in a. In the first step, we will draw the nucleus of the carbon atom. Web in this video we'll look at the atomic structure and bohr model for the carbon atom (c). We’ll use a bohr diagram to visually represent where the electrons are around the nucleus of the c. It belongs to group 14 of the periodic table. Web carbon. This figure explains the bonding in a co2 molecule. Each o atom starts out with six (red) electrons and c with four (black) electrons, and each bond behind an o atom and the c atom consists of two electrons from the o and two of the four electrons from the c. We’ll use a bohr diagram to visually represent where. In the first step, we will draw the nucleus of the carbon atom. Pick one of the bond types (single, double, triple, up, down) and add or modify bonds. Web so if we ignore hydrogen, the central atom is either going to be carbon or oxygen. All structures must begin with an atom. The quantum mechanical model of the atom. Oxygen is more electronegative than carbon. Web drawing every bond and every atom is tedious, however, so chemists have devised several shorthand ways for writing structures. It is nonmetallic and tetravalent —meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. Web so if we ignore hydrogen, the central atom is either going to be carbon or oxygen. Carbon is the first element that has precisely this shape, it is made up of 12 protons in a icosahedron shape. Web the most common carbon atoms have six protons and six neutrons in their nuclei. Click to place an atom. Web this image of a carbon atom is taken from a postsecondary chemistry textbook. Web carbon (from latin carbo 'coal') is a chemical element; Web how to draw atoms (bohr model) in this lesson i present an overview of: Carbon typically shares electrons to achieve a complete valence shell, forming bonds with multiple other atoms. Quantum numbers for the first four shells. Pick one of the fragments (benzene, cyclopropane, etc.) and add fragments. The periodic table, electron shells, and orbitals. It has symbol c and atomic number 6. Each o atom starts out with six (red) electrons and c with four (black) electrons, and each bond behind an o atom and the c atom consists of two electrons from the o and two of the four electrons from the c.REMC 22 / Figure App
3d render of atom structure of carbon isolated over white background
Carbon atom diagram hires stock photography and images Alamy
Diagram Of A Carbon Atom Illustration Getty Images
Carbon Atom Ascension Glossary
Carbon atomic structure (437243) Illustrations Design Bundles
Carbon Atom Diagram
Carbon Atom Molecular Structure Labels Stock Vector Illustration of
Carbon atom diagram concept Royalty Free Vector Image
Carbon atom diagram hires stock photography and images Alamy
Hydrogen Atoms Are Omitted But Are Assumed To Be Present To Complete Each Of Carbon's Four Bonds.
Web Carbon (C ), As A Group 14 Element, Has Four Electrons In Its Outer Shell.
The Atomic Number Symbolizes The Number Of Protons In An Atom.
Hydrogens That Are Attached To Elements Other Than Carbon Are Shown.
Related Post: